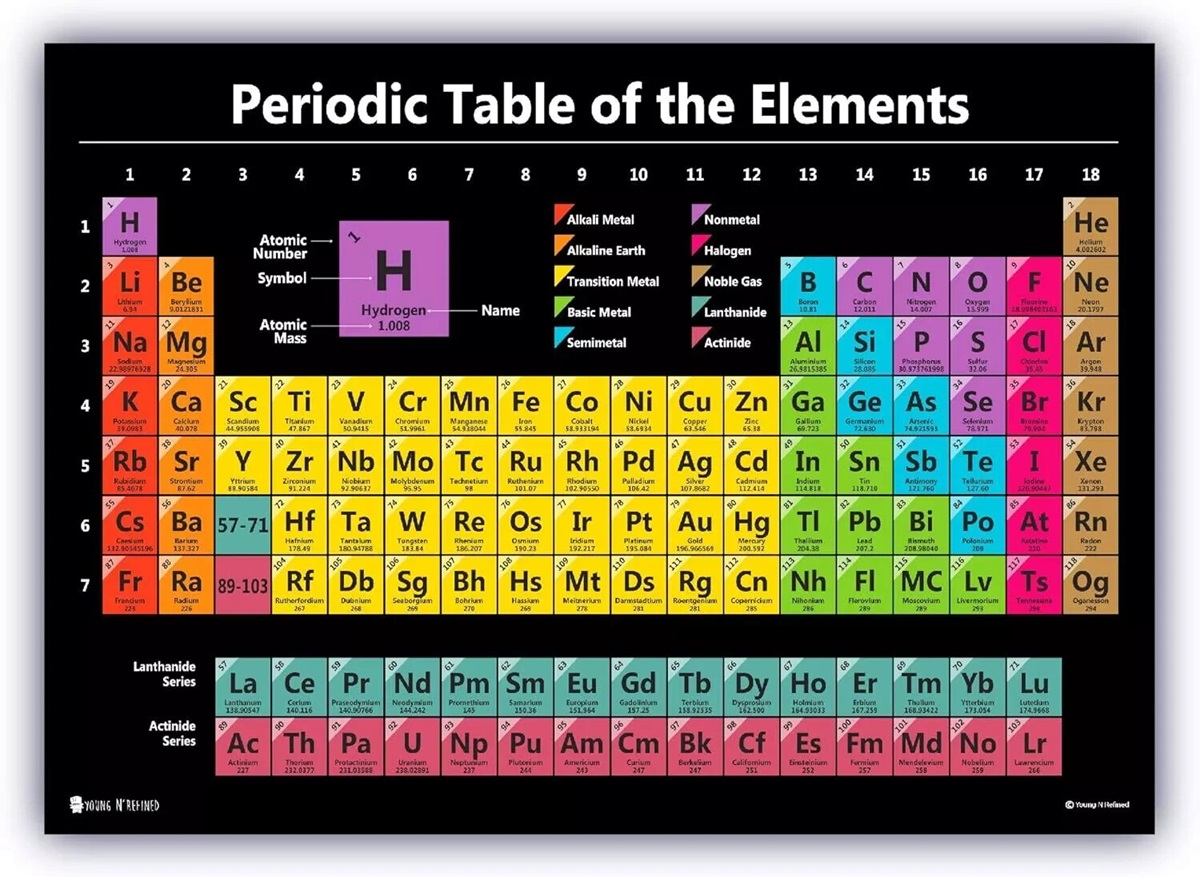
The Periodic Table of Elements: Origins, Modern Relevance, and the Elements Shaping Our Future
The periodic table of elements is one of the most powerful organizing tools in science. What began as a 19th-century attempt to classify known substances has evolved into a predictive framework that underpins chemistry, physics, biology, engineering, and modern technology. Today, the periodic table not only explains how matter behaves but also guides innovation in medicine, renewable energy, computing, and advanced materials science.
This article explores how the periodic table was developed, the newest elements added to it, the elements most critical in health, science, and technology, the economic value of key elements, and the origins of the short labels used to represent them.
The Origins of the Periodic Table
The modern periodic table is largely credited to Dmitri Mendeleev, who in 1869 arranged elements by increasing atomic mass and observed repeating (periodic) patterns in their chemical behavior. Mendeleev’s genius was not just organizing known elements but predicting the existence and properties of elements not yet discovered. When elements like gallium and germanium were later found and matched his predictions, the periodic table gained credibility.
Later, the work of Henry Moseley refined the system by organizing elements according to atomic number (number of protons), rather than atomic mass. This adjustment resolved inconsistencies and established the modern structure of the periodic table.
Today, the periodic table is maintained and standardized by the International Union of Pure and Applied Chemistry (IUPAC), which oversees element naming and classification.
How the Periodic Table Is Organized
The periodic table arranges elements by atomic number in rows (periods) and columns (groups). Elements within the same group share similar chemical properties due to similar electron configurations.
Major categories include:
• Alkali metals (highly reactive, e.g., sodium)
• Alkaline earth metals (magnesium, calcium)
• Transition metals (iron, copper, gold)
• Halogens (fluorine, chlorine)
• Noble gases (helium, neon)
• Lanthanides and actinides (rare earth and radioactive elements)
The structure allows scientists to predict how elements will bond, react, and behave under different conditions.
Newly Added Elements
The most recent additions to the periodic table were officially recognized in 2016. These include:
• Nihonium (Nh)
• Moscovium (Mc)
• Tennessine (Ts)
• Oganesson (Og)
These elements are synthetic, meaning they do not occur naturally and are created in particle accelerators. They are highly unstable and exist only for fractions of a second before decaying.
While they currently have limited practical application, their creation helps scientists understand nuclear stability and the theoretical “island of stability” — a predicted region where superheavy elements might exist longer.
Key Elements for the Future
As we look ahead, several elements stand out as strategically critical for global development.
1. Lithium (Li)
Lithium is essential for rechargeable batteries used in electric vehicles (EVs), smartphones, and renewable energy storage systems. As the world transitions to clean energy, lithium demand is surging.
2. Silicon (Si)
Silicon remains foundational to semiconductors and solar panels. It is the backbone of computing and digital technology.
3. Rare Earth Elements (Lanthanides)
Elements like neodymium and dysprosium are used in powerful magnets for wind turbines and EV motors.
4. Hydrogen (H)
Hydrogen is being explored as a clean fuel alternative. Green hydrogen production could significantly impact energy systems.
5. Uranium (U)
Uranium fuels nuclear reactors and may play a role in low-carbon energy expansion.
Elements Most Used in Health
Medicine relies on numerous elements:
• Carbon (C), Hydrogen (H), Oxygen (O), Nitrogen (N): Fundamental to organic molecules and life itself.
• Iron (Fe): Essential for hemoglobin and oxygen transport.
• Calcium (Ca): Critical for bone health and nerve function.
• Iodine (I): Vital for thyroid hormone production.
• Technetium-99m: Widely used in diagnostic imaging.
• Platinum (Pt): Used in chemotherapy drugs like cisplatin.
Additionally, elements like cobalt and chromium are used in joint replacements and medical implants due to strength and biocompatibility.
Elements Most Used in Science and Technology
Modern technology depends on specific high-performance elements:
• Copper (Cu): Electrical wiring and electronics.
• Gold (Au): Corrosion-resistant connectors in electronics.
• Silver (Ag): Conductivity and antimicrobial applications.
• Gallium (Ga): Semiconductors and LEDs.
• Titanium (Ti): Aerospace, medical implants, and strong lightweight alloys.
• Helium (He): Cryogenics and MRI machines.
Without these elements, much of modern life — smartphones, internet infrastructure, renewable energy systems — would not function.
Monetary Value of Key Elements
Element values fluctuate based on supply, demand, and geopolitical factors. Approximate trends include:
• Gold (Au): One of the most valuable and stable precious metals, typically priced in thousands of dollars per ounce. Used in jewelry, electronics, and as a financial reserve asset.
• Platinum (Pt): Valuable industrial metal used in catalytic converters and medicine.
• Palladium (Pd): Often even more expensive than gold due to automotive demand.
• Lithium (Li): Prices have surged due to EV battery demand.
• Rhodium (Rh): One of the rarest and most expensive metals, with prices that have spiked above gold due to limited supply.
Meanwhile, elements like iron and aluminum are inexpensive due to abundance and large-scale production.
How Did Elements Get Their Short Labels?
Each element has a one- or two-letter symbol. These short names often come from:
1. Latin Names
• Sodium = Na (from Natrium)
• Potassium = K (from Kalium)
• Iron = Fe (from Ferrum)
• Gold = Au (from Aurum)
2. Greek Roots
• Helium (He), from Helios (sun)
3. Named After Scientists
• Curium (Cm), after Marie and Pierre Curie
• Einsteinium (Es), after Albert Einstein
4. Named After Places
• Americium (Am), after America
• Californium (Cf), after California
• Nihonium (Nh), from Japan (Nihon)
The system of symbols was standardized to avoid confusion across languages. Latin-based symbols allowed international consistency.
The Future of the Periodic Table
Scientists continue exploring whether additional superheavy elements (119 and beyond) can be synthesized. If successful, they would expand the seventh period and possibly start an eighth.
Beyond discovering new elements, research focuses on:
• Sustainable extraction of rare earth metals
• Recycling critical minerals
• Developing alternative materials to reduce reliance on scarce elements
• Exploring extraterrestrial mining (e.g., asteroid resources)
As technology advances, the periodic table remains both a map of matter and a roadmap for innovation.
Conclusion
The periodic table is far more than a classroom chart — it is a dynamic framework that organizes the building blocks of the universe. From Mendeleev’s early predictions to modern synthetic elements like nihonium and oganesson, the table has grown alongside scientific discovery.
Certain elements — lithium, silicon, rare earth metals, hydrogen, uranium — will shape the future of energy and technology. Others, like carbon, iron, and calcium, remain fundamental to life and health. Precious metals such as gold, platinum, and palladium carry both industrial and monetary significance.
The short symbolic labels reflect a rich history of linguistic tradition, scientific honor, and global collaboration.
As humanity advances into an era of clean energy, AI-driven materials science, and possibly space exploration, the periodic table will remain central — a testament to our quest to understand and harness the fundamental elements of existence.

 3614
3614